Medical Equipment Electronics Manufacturing
PCBA and box build for medical equipment electronics — patient monitoring, diagnostic devices, and health tech assembled under ISO 9001 certified processes in Bangalore.
Medical-grade build discipline. Without exception.
Medical electronics — even non-implantable sub-assemblies — carry a quality expectation that most EMS environments cannot meet. A missed component on a patient monitor board or a cold joint on a diagnostic device PCBA is not a production defect; it is a patient safety risk. Vishesh builds medical equipment PCBA under the same zero-defect process discipline applied to our defense and automotive programmes.
India's medical device sector is growing at 12%+ CAGR, with domestic OEMs building patient monitoring, diagnostic, and health tech products for Indian healthcare and export. Vishesh handles medical equipment PCBA and box build under one quality system as an ISO certified EMS partner.
From first article to volume production — one facility, one quality system, one point of contact. Request a quote and we'll respond within one business day.
Zero-Defect Process Discipline
Controlled reflow, 100% AOI, and X-ray for BGA and hidden joints — the same build discipline as defense and automotive. No exceptions by programme size.
Component-Level Traceability
Every component tracked from incoming inspection to dispatch. Full traceability record for every batch — the audit trail medical equipment OEMs require.
ISO 9001 Quality System
Full PCBA and box build scope. One quality system from prototype to volume — no process discontinuity as programmes scale.
What We Build For
If you're building any of the following, Vishesh's facility and certifications make us a qualified manufacturing partner.
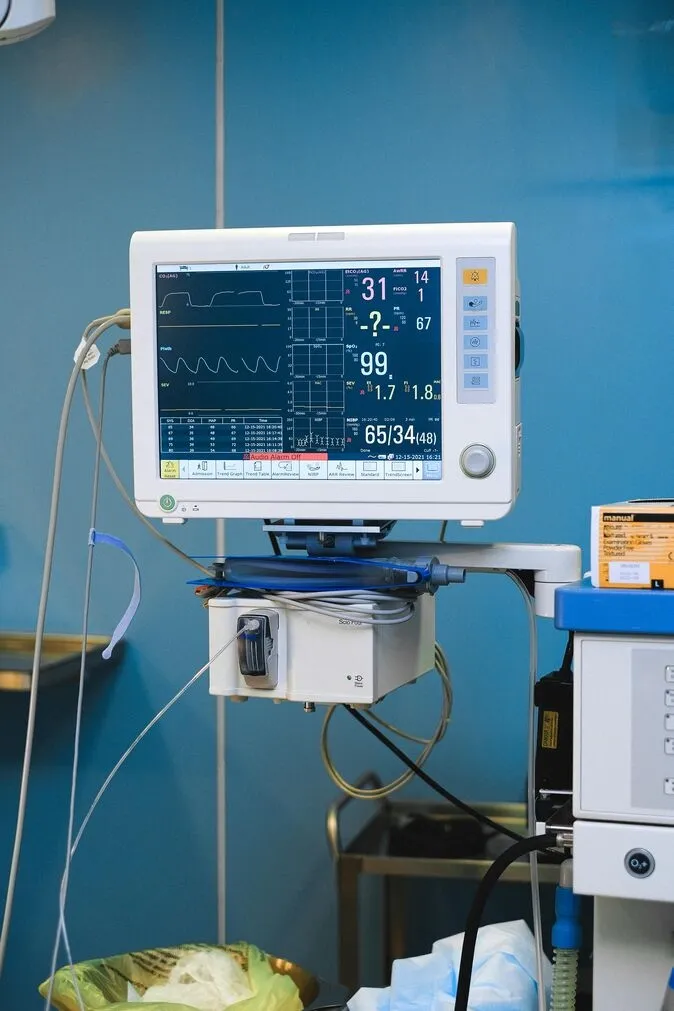
ECG monitor boards, pulse oximeter PCBAs, vital signs display assemblies.

Portable diagnostic device PCBAs, point-of-care testing equipment boards.
Nurse call system PCBAs, patient bed controller boards.

Wearable health monitor PCBAs, remote patient monitoring boards.
Imaging equipment interface boards, display controller PCBAs.
Infrastructure for Medical Equipment Electronics




What Vishesh Can Manufacture for Your Medical Equipment Programme
| Capability | Detail |
|---|---|
| SMT Lines | 3 lines, 206,700 CPH total |
| Component Range | 0201 to BGA, LGA, CSP, fine-pitch QFP |
| PCB Layers | Up to 16-layer |
| THT / Wave Soldering | 6,000 boards/day |
| Inspection | 100% AOI, X-ray (BGA/LGA), IPC-certified trainer on-site |
| Traceability | Component-level, incoming inspection to dispatch |
| Box Build | Full device integration |
| Quality Certification | ISO 9001:2015, IATF 16949:2016 (TÜV Rheinland) |
| Environmental | ISO 14001:2015, ISO 45001:2018 (TÜV NORD) |
| Facility | 45,000 sq ft, Bangalore |
Vishesh's Plant 1 handles all medical equipment PCBA — SMT assembly, AOI, and X-ray inspection. Plant 2 handles box build and full device integration for finished medical assemblies. Both plants operate under one quality system at our Bangalore facility.
Component-level traceability from incoming inspection to dispatch is standard — not optional — on every medical equipment build. For a full view of Vishesh's end-to-end EMS partner capability, visit our services overview.

Certified. Traceable. Audit-Ready.
ISO 9001:2015
Quality Management System
Certified by TÜV Rheinland. Valid to November 2028. Covers full PCBA and box build manufacturing operations at Vishesh's Bangalore facility. The quality management foundation of every medical equipment electronics programme — 100% AOI, X-ray, and component-level traceability built into process.
IATF 16949:2016
Automotive-Grade Process Discipline
Certified by TÜV Rheinland. Valid to November 2028. FMEA, APQP, and process control requirements applied to every production run — the same process rigour that benefits automotive programmes is available to medical equipment OEMs.
ISO 14001:2015
Environmental Management
Certified by TÜV NORD. Valid to November 2028. Environmental management system covering Vishesh's manufacturing operations — relevant for OEMs with sustainability requirements in their supply chain qualification.
ISO 45001:2018
Occupational Health & Safety
Certified by TÜV NORD. Valid to November 2028. Occupational health and safety management system. Demonstrates the operational maturity that responsible supply chain partners require from their manufacturers.
Frequently Asked Questions
What medical equipment electronics does Vishesh manufacture?
Vishesh assembles PCBAs and box builds for patient monitoring peripherals, diagnostic equipment, hospital infrastructure electronics, health tech wearables, and medical imaging support systems. These are non-implantable, non-life-critical electronic sub-assemblies and equipment boards.
Is Vishesh ISO certified for medical equipment manufacturing?
Vishesh holds ISO 9001:2015 certification from TÜV Rheinland, valid to November 2028. Our quality management system covers the full PCBA and box build scope with 100% AOI, X-ray inspection, and component-level traceability on every build.
What inspection processes does Vishesh use for medical equipment PCBA?
100% AOI on every SMT line using Kohyoung inspection systems, Unicomp X-ray for BGA and hidden joint verification, and component-level traceability from incoming inspection to dispatch. No board leaves uninspected.
Can Vishesh handle both prototype and production volumes for medical equipment OEMs?
Yes. Vishesh supports prototype builds, NPI, and volume production under one quality system. Process parameters and build knowledge carry forward from prototype to production without a supplier change.
Where is Vishesh located and what medical device segments does it serve?
Vishesh is based in Bangalore and serves medical equipment OEMs across patient monitoring, diagnostic equipment, hospital infrastructure, and health tech segments across India.
Get a quote for your medical equipment programme
Share your requirements. We'll respond within one business day.